cancer 201310 9
Aging (Albany NY). 2012 Dec;4(12):932-51.
Hubackova S, Krejcikova K, Bartek J, Hodny Z.
Department of Genome Integrity, Institute of Molecular Genetics, v.v.i., Academy of Sciences of the Czech Republic, Prague, Czech Republic.
Abstract
Many cancers arise at sites of infection and inflammation. Cellular senescence, a permanent state of cell cycle arrest that provides a barrier against tumorigenesis, is accompanied by elevated proinflammatory cytokines such as IL1, IL6, IL8 and TNFα. Here we demonstrate that media conditioned by cells undergoing any of the three main forms of senescence, i.e. replicative, oncogene- and drug-induced, contain high levels of IL1, IL6, and TGFb capable of inducing reactive oxygen species (ROS)-mediated DNA damage response (DDR). Persistent cytokine signaling and activated DDR evoke senescence in normal bystander cells, accompanied by activation of the JAK/STAT, TGFβ/SMAD and IL1/NFκB signaling pathways. Whereas inhibition of IL6/STAT signaling had no effect on DDR induction in bystander cells, inhibition of either TGFβ/SMAD or IL1/NFκB pathway resulted in decreased ROS production and reduced DDR in bystander cells. Simultaneous inhibition of both TGFβ/SMAD and IL1/NFκB pathways completely suppressed DDR indicating that IL1 and TGFβ cooperate to induce and/or maintain bystander senescence. Furthermore, the observed IL1- and TGFβ-induced expression of NAPDH oxidase Nox4 indicates a mechanistic link between the senescence-associated secretory phenotype (SASP) and DNA damage signaling as a feature shared by development of all major forms of paracrine bystander senescence.
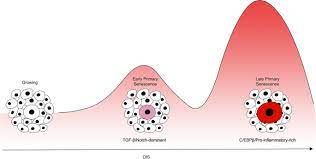
Mechanism of cytokine-induced bystander senescence. Primary senescent cells with persistently activated DNA damage response (DDR) signalling from unrepaired DNA lesions (stars) produce specific secretome (senescence-associated secretory phenotype; SASP) including more than two dosen of different cytokines. IL1beta and TGFbeta induce NADPH oxidase Nox4 in paracrine manner in normal neighboring cells. Nox4-generated reactive oxygen species (ROS) cause DNA damage response, which persistence activates secondary senescence with specific secondary SASP.
 Euro
Euro
 US Dollar
US Dollar
